A general strategy to enhance the alkaline stability of anion exchange membranes†
Abstract
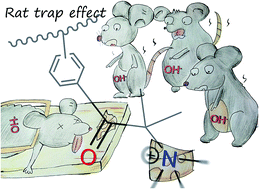
Quaternary ammonium (QA)-type polysulfone (PSf) anion exchange membranes (AEMs) with pendant carbonyl groups were synthesized by an environmentally friendly method. Low toxicity 3-chloro-2,2-dimethylpropionyl chloride (CDPC) was chosen as an electrophilic reagent to introduce chloroalkyl groups to PSf, instead of highly toxic chloromethyl methyl ether. The experimental conditions were carefully optimized to avoid the Friedel–Crafts alkylation side reaction on the chloroalkyl group. After quaternizing with trimethylamine (TMA), AEMs were prepared by solution casting. The obtained AEMs showed good ionic conductivity and mechanical stability. The alkaline stability of these AEMs was excellent; the AEMs can maintain 95% of their initial conductivity after immersion in a 1 M NaOH aqueous solution at 80 °C for 10 days. The reason for this outstanding alkaline stability was evaluated by NMR and density functional theory calculations. The most severe degradation pathway, the SN2 reaction, can be weakened by the competing reaction between OH− and carbonyl groups. The formed intermediate oxygen anion can decrease the possibility of SN2 by reducing the electro-static potential of the QA groups. Because of the reversibility of the reaction between OH− and carbonyl groups, QA groups can survive longer in strong alkaline conditions at high temperature. Also, the possibility of SN2 reaction on the aromatic polymer chain can be decreased because of the positive charge carrier, QA groups, which are not directly attached to benzyl groups. Hoffmann elimination can not happen because there was no hydrogen atom on the β-position of QA group. These structural advantages of the synthesized AEMs suggest another strategy that the potential for generalisation to other AEM systems and to improve the alkaline stability of QA-type AEMs.


 Please wait while we load your content...
Please wait while we load your content...