Nitrogen-doped porous graphene as a highly efficient cathodic electrocatalyst for aqueous organic redox flow battery application†
Abstract
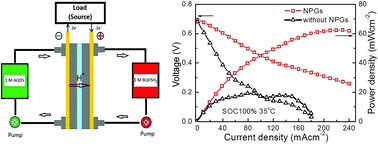
The redox flow battery (RFB) is considered one of the most attractive energy storage technologies because of its high efficiency, long service life and great safety. In this work, nitrogen-doped porous graphene (NPG) was achieved by embedding nitrogen atoms into the graphitic matrix via a sacrificial-template-assisted pyrolysis approach, and then was employed to catalyze electrochemically the redox reaction of 1,2-dihydrobenzoquinone-3,5-disulphonic acid (BQDSH2) for aqueous organic RFB application. The electrocatalytic performances of this catalyst toward the BQDS/BQDSH2 redox couple were greatly enhanced, including a 6.7 times higher rate constant and much lower peak potential separation value compared to those of a glassy carbon electrode. Furthermore, a 9,10-anthraquinone-2,7-disulfonic acid (AQDS)/BQDS RFB with NPGs as the cathodic BQDS/BQDSH2 catalyst exhibited a maximum power density of ca. 62.4 mW cm−2, about 3.3 times higher than that of the pristine RFB without NPGs, and a minimal discharge capacity fade during the cycling test. The abundant nitrogen defects in NPGs are beneficial to the BQDS/BQDSH2 redox reaction by providing active sites and enhancing the transfer rate of protons and reactants, contributing to the improvement of AQDS/BQDS RFB performance.


 Please wait while we load your content...
Please wait while we load your content...