Computational screening for high-activity MoS2 monolayer-based catalysts for the oxygen reduction reaction via substitutional doping with transition metal†
Abstract
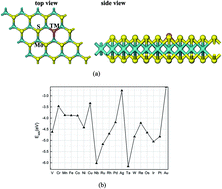
Rational design of non-noble materials as highly efficient, economical, and durable platinum-free oxygen reduction reaction (ORR) electrocatalysts is crucial to the large-scale application of fuel cells. In this work, using density functional theory (DFT) computations, the catalytic activity for the ORR of substitutionally doped MoS2 monolayers with various transition metal (TM) atoms was systemically explored. The computations reveal that these TM atoms can strongly interact with the S-vacancy and significantly modify the electronic and magnetic properties of MoS2 monolayer. On the basis of the computed free energies, it is evident that Cu-embedded MoS2 monolayer exhibits the best ORR catalytic activity with the minimum overpotential of 0.63 V because of its optimal interaction with ORR species. Thus, this study suggests that substituted doping with a suitable transition metal is an effective method to improve the catalytic activity of MoS2 monolayer for the ORR, which could be useful in development of novel 2D ORR electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...