Enhanced hydrogen storage properties of MgH2 with numerous hydrogen diffusion channels provided by Na2Ti3O7 nanotubes†
Abstract
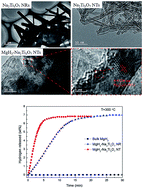
Na2Ti3O7 nanotubes (NTs) with a uniform diameter of 10 nm and Na2Ti3O7 nanorods (NRs) with a diameter of 100–500 nm were synthesized via a hydrothermal method and a solid-state method, respectively, and then introduced into MgH2 by ball milling to catalyze the hydrogenation/dehydrogenation process. The MgH2–Na2Ti3O7 NT and MgH2–Na2Ti3O7 NR composites can desorb 6.5 wt% H2 within 6 min and 16 min at 300 °C, respectively, while the bulk MgH2 hardly releases any hydrogen even over a much longer time. In addition, isothermal rehydrogenation measurements show that the MgH2–Na2Ti3O7 NT composite can absorb 6.0 wt% H2 within 60 s at 275 °C and can even absorb 1.5 wt% H2 within 30 min at a temperature as low as 50 °C. TEM and HRTEM analyses indicate that the Na2Ti3O7 NTs are homogeneously distributed in MgH2, which catalyze the de-/rehydrogenation of MgH2 and meanwhile offer numerous diffusion channels to significantly accelerate the transportation of hydrogen atoms. Moreover, compared with bulk MgH2 and the MgH2–Na2Ti3O7 NR composite, the activation energy of the MgH2–Na2Ti3O7 NT composite is significantly decreased to 70.43 kJ mol−1. Such Na2Ti3O7 NTs with a unique morphology of the catalyst being distributed as nanotubes in MgH2 are believed to pave the way for the future design of hydrogen storage materials with excellent hydrogen storage performances.


 Please wait while we load your content...
Please wait while we load your content...