Computational design of cobalt-free mixed proton–electron conductors for solid oxide electrochemical cells†
Abstract
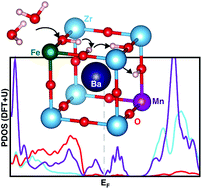
Proton-conducting solid-oxide electrolyzer and fuel cells (PC-SOECs/FCs) represent viable, intermediate-temperature green technologies for H2 production and conversion. While PC ceramics have been extensively investigated as electrolytes for PC-SOECs/FCs, the development of corresponding single-phase electrode components has been hindered by difficulties in finding efficient mixed proton–electron conductors (MPECs), with also effective catalytic activity toward oxygen reduction and evolution reactions (ORR/OER). To address this challenge, we applied first-principles methods (PBE+U) to design new perovskite-oxide MPEC electrodes based on the known BaZrO3 PC ceramic. Our strategy has been to modify the parent material by substituting Zr with earth abundant transition metals, namely Mn and Fe. We found Zr : Mn and Zr : Fe ratios of 0.75 : 0.25 to be sufficient to obtain electronic structural features that can enable electric conductivity. We also investigated other relevant processes for MPEC-based electrodes: hydration, proton migration, and ORR/OER electrocatalysis. From calculations of key descriptors associated with these processes, we found that Zr substitution with Mn or Fe delivers in both cases promising PC-SOEC/FC electrodes. Moreover, our first-principles results highlight the specific qualities of Mn and Fe: the first provides better electronic features and electrocatalytic activities, whereas the latter allows for better hydration and proton migration. In perspective, our findings present clear indications for the experimental implementation and test of new low-cost materials for solid-oxide electrochemical cells.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...