Sn4+ self-doped hollow cubic SnS as an efficient visible-light photocatalyst for Cr(vi) reduction and detoxification of cyanide†
Abstract
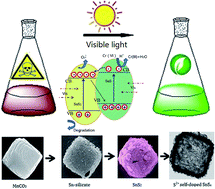
Sn4+ self-doped hollow SnS microcubes are controllably designed by a simple template-assisted synthesis followed by reduction under low temperature. The in situ self-doping enhances the interaction between SnS2 and SnS. The 3D hollow structures of the photocatalyst improve the light efficiency significantly due to effective multiple reflections in the cave. Therefore, this novel material shows a remarkable reduction performance for Cr(VI) and detoxification of cyanide under visible light irradiation. The concentration of Cr(VI) decreases sharply with the irradiation time, and 99.6% of Cr(VI) is degraded within 50 min. Meanwhile, the chromium ions can be adsorbed completely. Moreover, the removal rate for cyanide is as high as 97.2%. Combined with the analysis of in situ ATR-FTIR spectroscopy technology, the photocatalytic reduction mechanism of Cr(VI) is confirmed as a proton coupled electron transfer process. The improved photocatalytic activity of the Sn4+ self-doped SnS can be attributed to the enhanced photoabsorption properties and effective separation of photoinduced charge carriers. This general strategy provides a novel route for the synthesis of hollow cubic structural materials, which can be used for photocatalysis, energy storage and wastewater purification applications.


 Please wait while we load your content...
Please wait while we load your content...