A microwave-assisted synthesis of CoO@Co core–shell structures coupled with N-doped reduced graphene oxide used as a superior multi-functional electrocatalyst for hydrogen evolution, oxygen reduction and oxygen evolution reactions†
Abstract
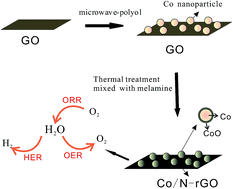
A novel electrochemical catalyst comprising CoO@Co nanoparticles with a core–shell structure immobilized on N-doped reduced graphene oxide (rGO) (CoO@Co/N-rGO) has been synthesized using a convenient and controllable technique — combining a rapid microwave-polyol method with a vacuum thermal treatment. Excellent features including the Co/N-doping effect, introduction of CoO@Co particles with a core–shell structure and good contact between the CoO@Co particles and N-doped graphene result in a highly multi-functional catalytic efficiency. The catalyst exhibits remarkable catalytic activity and superior stability towards the hydrogen evolution reaction, offering a low overpotential of 140 mV for 10 mA cm−2 in 0.5 M H2SO4 and 237 mV in 0.1 M KOH. The catalyst also shows excellent oxygen reduction reaction activity in 0.1 M KOH, a similar four-electron pathway, which is comparable to that of a commercial Pt/C catalyst, and superior stability. In addition, a good electrochemical performance towards the oxygen evolution reaction was observed for the catalyst, achieving a current density of 10 mA cm−2 with a small overpotential of 1.67 V in 0.1 M KOH, which is comparable to that of a commercial RuO2 catalyst. The unusual catalytic activities arise from the synergetic chemical coupling effects of metallic Co, cobalt oxides and Co/N-doped graphene. This study provides a new attractive multi-functional catalyst material for unitized regenerative fuel cells and overall water splitting technologies.


 Please wait while we load your content...
Please wait while we load your content...