A two-volt aqueous supercapacitor from porous dehalogenated carbon†
Abstract
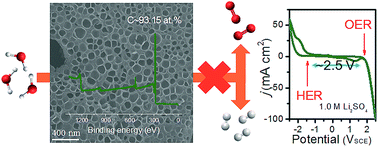
Aqueous supercapacitors are among the most promising materials for clean and renewable energy storage; however, their utilization for applications is limited by their low specific energy arising from issues due to water splitting. Herein, water splitting-inactive electrode materials of mesoporous carbon were fabricated through the dehalogenation of polyvinylidene fluoride (PVDF) by sodium ethoxide (EtONa). The as-generated EtOH and NaF were demonstrated to facilitate the formation of hierarchical porous carbon based on the bubble and templating effects. Due to full dehalogenation, the resulting carbon materials acquired high contents of carbon without unfavorable alien dopants. The resulting porous, dehalogenated, carbon materials were applied as electrode materials for an aqueous supercapacitor neutral electrolyte. The significantly suppressed water-splitting activity has been addressed in the electrochemical system. Therefore, the open circuit voltage (OCV) could be safely expanded to 2.0 V. Over 94.0% of the capacitance was also maintained after cycling 5000 times at 5.0 A g−1. Our strategy that utilizes water-splitting inactive electrode materials to expand the working window of an aqueous supercapacitor may enable the design of materials for aqueous supercapacitors with higher OCVs over 2.0 V.


 Please wait while we load your content...
Please wait while we load your content...