Distinct nanoscale reaction pathways in a sulfide material for sodium and lithium batteries†
Abstract
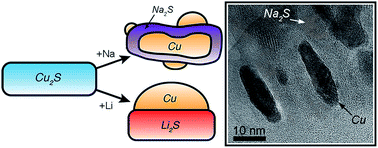
Sodium-ion batteries are attractive because of their potentially lower cost than lithium-ion systems. However, the larger ionic radius of Na+ means that candidate electrode materials often undergo more substantial volumetric changes during reaction as compared to Li-ion batteries, and these changes must be understood and controlled for the development of electrode materials with long cycle life. Here, nanoscale-to-macroscale transformation pathways are investigated in real time in Cu2S (a sulfide electrode material) during electrochemical reaction with Na and Li. In situ and ex situ X-ray diffraction reveal that the overall phase transformations in Cu2S electrodes are similar within both Na and Li cells. However, in situ transmission electron microscopy (TEM) shows that the nanoscale reaction pathways differ significantly, which likely contributes to observed differences in electrochemical behavior. Despite these dissimilarities, Na/Cu2S electrochemical cells are shown to exhibit excellent cycle life for the first time (negligible capacity decay over 400 cycles), which is similar to the Li case. Thus, although the more substantial volume changes during the sodiation of Cu2S induce a new reaction pathway, they do not cause accelerated capacity decay, as is commonly argued for Na-ion materials. These results suggest that other large-volume-change electrode materials may also be engineered for long cycle life in next-generation Na-ion batteries.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...