Self-assembly behaviors of dibenzylidene sorbitol hybrid organogels with inorganic silica
Abstract
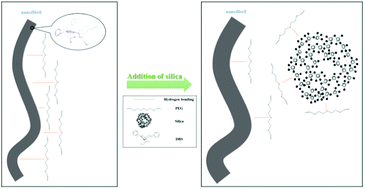
Molecular interactions, rheological behaviors and microstructures of 1,3:2,4-dibenzylidene-D-sorbitol (DBS)/poly(ethylene glycol) (PEG) organogel–inorganic silica hybrid materials are discussed in this study. DBS can dissolve in low-molecular-weight PEG to form organogels. The self-assembly behavior of these organogels was significantly influenced by the addition of the inorganic silica. The π interactions between the phenyl rings of DBS were not influenced by silica addition; however, the addition of silica affected the intermolecular hydrogen bonding of DBS, which interacts with PEG. The silica more likely interacted with PEG and decreased the intermolecular interactions between DBS and PEG, which resulted in an increase in the self-assembly of DBS. Therefore, the gel formation time and gel dissolution temperature increased as the amount of silica increased, as determined by dynamic rheological instruments. In addition, these organogel systems were all found to exhibit spherulite-like textures under polarized optical microscopy. The addition of silica and the increased DBS self-assembly in PEG resulted in a higher self-assembly temperature of the organogels. The higher temperature resulted in the presence of fewer nucleation sites and larger spherulite sizes in these systems. Small-angle X-ray scattering results demonstrated lamellar packing in these spherulite-like morphologies. Furthermore, the organogels with silica affected the intermolecular hydrogen bonding between DBS and PEG to facilitate the self-assembly of DBS, which resulted in increased diameter sizes of the DBS nanofibrils, as observed using scanning electron microscopy. It was observed that the silica was entrapped within these nanofibrillar networks.


 Please wait while we load your content...
Please wait while we load your content...