Controlled hierarchical assembly of gold nanoparticles in macroscopic films: from densely packed monolayers to networks of micropores and nanobundles†
Abstract
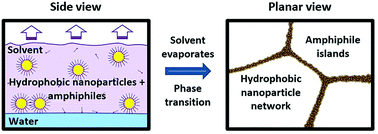
The present study demonstrates the ability of excess, weakly amphiphilic n-alkanethiols (n = 4, 12, 18) and solvent composition to tune through a wide range of large-scale, macroscopic architectures formed by alkanethiol-capped Au nanoparticles (NPs). Both the alkanethiols and NPs are significantly hydrophobic species and compete for surface area at an air–water interface. When solutions of the two species are spread on a large (50 cm2) water surface in a Teflon well, a thin film forms and exhibits co-existing macroscopic regions with various distinct NP self-assembled architectures, namely a close packed monolayer, a network phase characterized by micron-sized pores (micropores) surrounded by quasi-linear bundles of nanoparticles, and finally aggregates. We hypothesize that the co-existence of various NP architectures results from fast, non-uniform evaporation across the large water surface. When solutions are instead deposited on a smaller (5 cm2) water surface contained within a Teflon ring to control the water surface curvature and the evaporation rate is slowed, we show for the first time that NPs form macroscopically uniform self-assemblies whose architectures can be tuned from monolayers → monolayers with micropores → extended micropore/NP bundle networks by varying excess alkanethiol concentration and solvent composition. We propose that competition between NPs and excess alkanethiols for water surface area, and alkanethiol self-assembly as well as solvent dewetting play important roles in the formation of the network phase, and discuss a potential mechanism for its formation.


 Please wait while we load your content...
Please wait while we load your content...