Enhanced oxygen evolution on visible light responsive TaON photocatalysts co-loaded with highly active Ru species for IO3− reduction and Co species for water oxidation†
Abstract
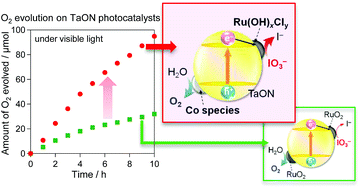
Loading an appropriate cocatalyst significantly enhances the activity of semiconductor photocatalysts in both conventional one-step water splitting and Z-scheme-type water splitting with a redox couple. In the present study, we examined the catalytic activity of Ru-based cocatalysts for IO3− reduction to improve the efficiency of O2 evolution on a TaON photocatalyst under visible light with an IO3− acceptor, which is an important half component in Z-scheme water splitting systems. X-ray photoelectron spectroscopy analyses and electrochemical measurements revealed that the calcination temperature in the loading of Ru species from aqueous RuCl3 significantly affected the catalytic activity for the reduction of IO3−. Calcination at or below 200 °C produced Ru(OH)xCly species that drastically enhanced the reduction of IO3−, whereas that at 300 °C generated conventional RuO2 that showed lower activity for IO3− reduction than Ru(OH)xCly but considerably enhanced the water oxidation. Co-loading the Ru(OH)xCly and Co cation species as water oxidation cocatalysts significantly improved O2 evolution on the TaON photocatalyst by more than a factor of two compared to that loaded with only the conventional RuO2 cocatalyst. This suggests the effectiveness of co-loading two different cocatalysts that are independently optimized for reduction and oxidation.
- This article is part of the themed collection: 2017 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...