Photoelectrochemical H2 evolution using TiO2-coated CaFe2O4 without an external applied bias under visible light irradiation at 470 nm based on device modeling†
Abstract
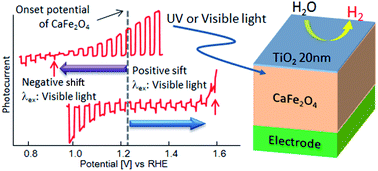
CaFe2O4 (CFO) can be used as a photocathode to evolve H2 from water in a photoelectrochemical cell. However, CFO degrades during operation and an external voltage is necessary for PEC H2 evolution because the onset potential is less than the potential required for water oxidation considering the overpotential at the counter electrode. In order to develop a reliable CFO electrode with a greater onset potential, improvement of chemical stability and suppression of surface recombination is necessary. In this study, a chemically stable electrode structure with a greater onset potential was achieved by coating the [00l]-oriented CFO with a thin layer of titanium dioxide (TiO2). A CFO|TiO2 electrode was designed using a device simulator. The simulation results predict that coating CFO with TiO2 produces a positive or negative shift in the onset potential under visible and ultraviolet light irradiation, respectively. The experimental onset potentials matched the simulation prediction. The observed onset potential for TiO2-coated CFO was around (1.6 V vs. RHE) under visible light (470 nm) and 0.9 V under ultraviolet light (300 nm), compared to 1.2–1.3 V vs. RHE for a bare CFO electrode. The onset potential (1.6 V) under visible light irradiation is the most positive onset potential among the oxide photocathodes ever reported for PEC water splitting. Using the TiO2-coated CFO as the photocathode and RuO2-loaded Pt as the anode, stable photocurrent was observed under 470 nm excitation without an external voltage and evolution of H2 from the system was confirmed.


 Please wait while we load your content...
Please wait while we load your content...