Rapid formation of self-organised Ag nanosheets with high efficiency and selectivity in CO2 electroreduction to CO†
Abstract
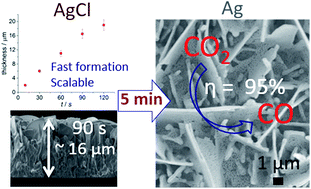
Rapid and scalable production of electrocatalysts with high conversion efficiency and product selectivity are essential for practical application of electrochemical CO2 reduction. Here we report highly efficient and selective free-standing silver (Ag) nanosheet-based electrocatalysts that were produced in less than 10 min via an electrochemical oxidative–reductive approach. The hierarchical structures of Ag nanosheets provide an enhanced surface area and favourable gas transport/diffusion. The interconnected nanosize Ag (∼45 ± 10 nm) sheets produce among the best performance for aqueous CO2 to CO reduction for this class of materials. The conversion selectivity was approximately 95% at an overpotential as low as 0.29 V. This rational experimental design strategy may inspire efforts towards developing rapidly synthesized and efficient CO2 reduction electrocatalysts.
- This article is part of the themed collection: RACI100: Celebrating Australian Chemistry


 Please wait while we load your content...
Please wait while we load your content...