Seawater operating bio-photovoltaic cells coupling semiconductor photoanodes and enzymatic biocathodes†
Abstract
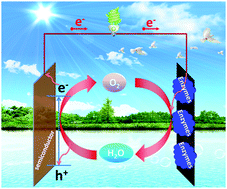
Access to fresh water and energy is ranked as one of the most severe challenges to humankind. The restricted availability of fossil fuels and clean water does not match the increasing energy demands and growing population needs, which, desirably, should be satisfied in the most sustainable, clean and inexpensive way. Here, we report clean and sustainable conversion of solar energy into electricity by photo- and bio-electrocatalytic recycling of the H2O/O2 redox couple in a hybrid bio-photovoltaic (BPV) membraneless cell comprising a sunlight-illuminated water-oxidizing semiconductor anode (either Zn-doped hematite or TiO2) and an oxygen-reducing enzymatic biocathode, in such environmental media as seawater. Upon simulated solar light illumination (AM 1.5G, 100 mW cm−2), the maximum power density (Pmax) generated by the cell was 236 and 21.4 μW cm−2 in 1 M Tris–HCl and seawater, both at pH 8, respectively. In seawater its ionic content inhibited mostly the activity of the photoanode, but not that of the biocathode. The obtained Pmax values were orders of magnitude higher than those of a photo-electrochemical cell with a Pt mesh cathode (0.32 μW cm−2 in seawater). The demonstrated thermodynamically feasible coupling of cost-effective photoactive materials such as TiO2 or hematite semiconductors and enzymatic counterparts in seawater media opens a prospective clean and sustainable way of transformation of the most abundant, clean and renewable source of energy – solar light – and the Earth's most massive water resource – seawater – into electricity, which can also be used for fresh water production.


 Please wait while we load your content...
Please wait while we load your content...