Molecular heterogeneous catalysts derived from bipyridine-based organosilica nanotubes for C–H bond activation†
Abstract
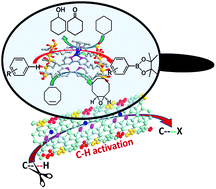
Heterogeneous metal complex catalysts for direct C–H activation with high activity and durability have always been desired for transforming raw materials into feedstock chemicals. This study described the design and synthesis of one-dimensional organosilica nanotubes containing 2,2′-bipyridine (bpy) ligands in the framework (BPy-NT) and their post-synthetic metalation to provide highly active and robust molecular heterogeneous catalysts. By adjusting the ratios of organosilane precursors, very short BPy-NT with ∼50 nm length could be controllably obtained. The post-synthetic metalation of bipyridine-functionalized nanotubes with [IrCp*Cl(μ-Cl)]2 (Cp* = η5-pentamethylcyclopentadienyl) and [Ir(cod)(OMe)]2 (cod = 1,5-cyclooctadiene) afforded solid catalysts, IrCp*-BPy-NT and Ir(cod)-BPy-NT, which were utilized for C–H oxidation of heterocycles and cycloalkanes as well as C–H borylation of arenes. The cut-short nanotube catalysts displayed enhanced activities and durability as compared to the analogous homogeneous catalysts and other conventional heterogeneous catalysts, benefiting from the isolated active sites as well as the fast transport of substrates and products. After the reactions, a detailed characterization of Ir-immobilized BPy-NT via TEM, SEM, nitrogen adsorption, UV/vis, XPS, and 13C CP MAS NMR indicated the molecular nature of the active species as well as stable structures of nanotube scaffolds. This study demonstrates the potential of BPy-NT with a short length as an integration platform for the construction of efficient heterogeneous catalytic systems for organic transformations.
- This article is part of the themed collection: In celebration of Chinese New Year


 Please wait while we load your content...
Please wait while we load your content...