Total chemical synthesis of SUMO-2-Lys63-linked diubiquitin hybrid chains assisted by removable solubilizing tags†
Abstract
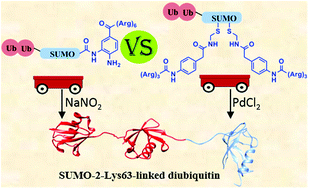
Small ubiquitin like modifier (SUMO) proteins are known to regulate many important cellular processes such as transcription and apoptosis. Recently, hybrid SUMO-ubiquitin chains containing SUMO-2 linked to Lys63-di-ubiquitin were found to play a major role in DNA repair. Despite some progress in understanding the role of these hybrid chains in DNA repair, there are various fundamental questions remaining to be answered. To further investigate the importance of hybrid SUMO-ubiquitin chains in DNA repair, the homogenous material of these chains, and their unique analogues, are needed in workable quantities. By applying advanced chemical strategies for protein synthesis, we report the first total chemical synthesis of four different SUMO-2-Lys63-linked di-ubiquitin hybrid chains, in which the di-ubiquitin is linked to different lysines in SUMO. In these syntheses, the usefulness of removable solubilizing tags is demonstrated, and two different approaches were examined in terms of reliability and efficiency. In the first approach, a poly-Arg tag was attached to the C-terminus of SUMO via a 3,4-diaminobenzoic acid cleavable linker, whereas in the second we attached the tag via a phenylacetamidomethyl linker, which can be cleaved by PdCl2. The comparison between these different strategies offers guidelines for future scale-up preparation of these analogues and other proteins, which currently use synthetic peptide intermediates that are difficult to handle and purify. The availability of the SUMO-ubiquitin hybrid chains opens up new opportunities for studying the role of these chains in DNA repair and other cellular processes.


 Please wait while we load your content...
Please wait while we load your content...