Halogenation of glycopeptide antibiotics occurs at the amino acid level during non-ribosomal peptide synthesis†
Abstract
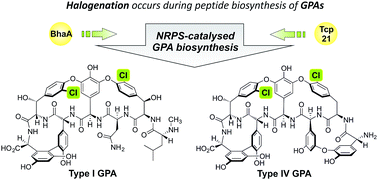
Halogenation plays a significant role in the activity of the glycopeptide antibiotics (GPAs), although up until now the timing and therefore exact substrate involved was unclear. Here, we present results combined from in vivo and in vitro studies that reveal the substrates for the halogenase enzymes from GPA biosynthesis as amino acid residues bound to peptidyl carrier protein (PCP)-domains from the non-ribosomal peptide synthetase machinery: no activity was detected upon either free amino acids or PCP-bound peptides. Furthermore, we show that the selectivity of GPA halogenase enzymes depends upon both the structure of the bound amino acid and the PCP domain, rather than being driven solely via the PCP domain. These studies provide the first detailed understanding of how halogenation is performed during GPA biosynthesis and highlight the importance and versatility of trans-acting enzymes that operate during peptide assembly by non-ribosomal peptide synthetases.


 Please wait while we load your content...
Please wait while we load your content...