Multi-electron reduction of sulfur and carbon disulfide using binuclear uranium(iii) borohydride complexes†
Abstract
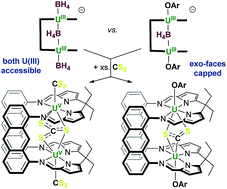
The first use of a dinuclear UIII/UIII complex in the activation of small molecules is reported. The octadentate Schiff-base pyrrole, anthracene-hinged ‘Pacman’ ligand LA combines two strongly reducing UIII centres and three borohydride ligands in [M(THF)4][{U(BH4)}2(μ-BH4)(LA)(THF)2] 1-M, (M = Li, Na, K). The two borohydride ligands bound to uranium outside the macrocyclic cleft are readily substituted by aryloxide ligands, resulting in a single, weakly-bound, encapsulated endo group 1 metal borohydride bridging the two UIII centres in [{U(OAr)}2(μ-MBH4)(LA)(THF)2] 2-M (OAr = OC6H2tBu3-2,4,6, M = Na, K). X-ray crystallographic analysis shows that, for 2-K, in addition to the endo-BH4 ligand the potassium counter-cation is also incorporated into the cleft through η5-interactions with the pyrrolides instead of extraneous donor solvent. As such, 2-K has a significantly higher solubility in non-polar solvents and a wider U–U separation compared to the ‘ate’ complex 1. The cooperative reducing capability of the two UIII centres now enforced by the large and relatively flexible macrocycle is compared for the two complexes, recognising that the borohydrides can provide additional reducing capability, and that the aryloxide-capped 2-K is constrained to reactions within the cleft. The reaction between 1-Na and S8 affords an insoluble, presumably polymeric paramagnetic complex with bridging uranium sulfides, while that with CS2 results in oxidation of each UIII to the notably high UV oxidation state, forming the unusual trithiocarbonate (CS3)2− as a ligand in [{U(CS3)}2(μ-κ2:κ2-CS3)(LA)] (4). The reaction between 2-K and S8 results in quantitative substitution of the endo-KBH4 by a bridging persulfido (S2)2− group and oxidation of each UIII to UIV, yielding [{U(OAr)}2(μ-κ2:κ2-S2)(LA)] (5). The reaction of 2-K with CS2 affords a thermally unstable adduct which is tentatively assigned as containing a carbon disulfido (CS2)2− ligand bridging the two U centres (6a), but only the mono-bridged sulfido (S)2− complex [{U(OAr)}2(μ-S)(LA)] (6) is isolated. The persulfido complex (5) can also be synthesised from the mono-bridged sulfido complex (6) by the addition of another equivalent of sulfur.


 Please wait while we load your content...
Please wait while we load your content...