Regio- and stereospecific rhodium-catalyzed allylic alkylation with an acyl anion equivalent: an approach to acyclic α-ternary β,γ-unsaturated aryl ketones†
Abstract
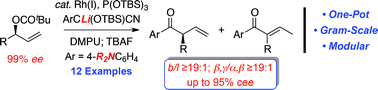
The regio- and stereospecific rhodium-catalyzed allylic alkylation of secondary allylic carbonates with cyanohydrin pronucleophiles facilitates the direct construction of acyclic α-ternary β,γ-unsaturated aryl ketones. Interestingly, this study illustrates the impact of deaggregating agents on regiocontrol and the electronic nature of the aryl component to suppress olefin isomerization. In addition, we demonstrate that the dimethylamino substituent, which modulates the pKa of the α-ternary β,γ-unsaturated aryl ketone, provides a useful synthetic handle for further functionalization via Kumada cross-coupling of the aryl trimethylammonium salt. Finally, the stereospecific alkylation of a chiral nonracemic secondary allylic carbonate affords the enantioenriched α-ternary aryl ketone, which was employed in a formal synthesis of trichostatic acid to illustrate that the allylic alkylation proceeds with net retention of configuration.


 Please wait while we load your content...
Please wait while we load your content...