Toward redesigning the PEG surface of nanocarriers for tumor targeting: impact of inner functionalities on size, charge, multivalent binding, and biodistribution†
Abstract
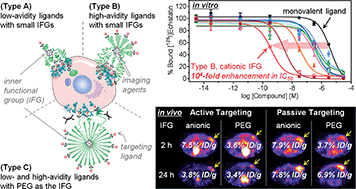
Achieving accurate and efficacious tumor targeting with minimal off-target effects is of paramount importance in designing diagnostic and therapeutic agents for cancer. In this respect, nanocarriers have gained enormous popularity because of their attainable multifunctional features, as well as tumor-targeting potential by extravasation. However, once administered into the bloodstream, nanocarriers face various in vivo obstacles that may significantly impair their performance needed for clinical translation. Herein, we demonstrate a strategy to enhance tumor-targeting efficiency by embedding functionalities in the interior region of partially PEGylated nanocarriers (ca. 10 nm in diameter), intended for active or passive targeting. The cooperative impact of these topologically inner functional groups (IFGs) was marked: enhancements of >100-fold in IC50in vitro (e.g., a high-avidity ligand with cationic IFGs) and >2-fold in tumor accumulation at 2 h post-injection in vivo (e.g., a high-avidity ligand with anionic IFGs), both against the fully PEGylated counterpart. Analogous to allosteric modulators, properly employed IFGs may substantially improve the process of effectively directing nanocarriers to tumors, which is otherwise solely dependent on avidity or extravasation.


 Please wait while we load your content...
Please wait while we load your content...