Continuous-flow synthesis of fluorine-containing fine chemicals with integrated benchtop NMR analysis†
Abstract
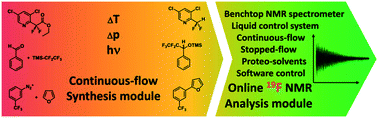
A compact lab plant was designed for the continuous-flow synthesis of fluorine-containing compounds and was combined with an NMR analysis platform based on a benchtop NMR spectrometer. The approach of a unified synthesis and analysis strategy for fine chemicals was applied to three different reactions, all employing fluorine as a chemical probe for online-19F NMR analysis. A high temperature synthesis for the deprotection of a CF2H group was done as well as Ruppert–Prakash reactions for the perfluoroalkylation of benzaldehyde as a model substrate. The C–H arylation of furan with a trifluoromethylated aryldiazonium salt was performed as an example of a photochemically catalyzed reaction. All three reaction classes challenge the synthesis and analysis setup differently according to sample preparation (premagnetization of bubble-free sample) and spectrometer sensitivity (signal to noise ratio, spectral resolution, scan number, substrate concentration and flow rate), but nonetheless prove the successful application of the continuous-flow synthesis of fluorinated fine chemicals with integrated online NMR analysis.


 Please wait while we load your content...
Please wait while we load your content...