Energetics of cellulose and cyclodextrin glycosidic bond cleavage†
Abstract
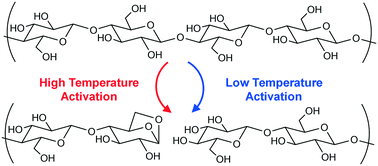
Thermochemical conversion of lignocellulosic materials for production of biofuels and renewable chemicals utilizes high temperature to thermally decompose long-chain cellulose to volatile organic compounds. Cellulose undergoes two distinct kinetic regimes of intra-chain scission: low-temperature glycosidic bond cleavage (T < 467 °C) associated with a low apparent activation energy and high-temperature glycosidic bond cleavage (T > 467 °C) associated with a high apparent activation energy. In this work, the initial breakdown kinetics of cellulose were examined from 385 °C to 505 °C using a millisecond, thin-film reactor called PHASR (pulse-heated analysis of solid/surface reactions). Using the cellulose surrogate, α-cyclodextrin, the energetics of each kinetic regime were characterized by measuring the conversion between 20 ms and 2.0 seconds. The low temperature kinetic regime exhibited glycosidic bond cleavage (Ea,1 = 23.2 ± 1.9 kcal mol−1, k0,1 = 2.0 × 107 s−1), while the high temperature kinetic regime (Ea,2 = 53.7 ± 1.1 kcal mol−1, k0,2 = 2.4 × 1016 s−1) was consistent with four reaction mechanisms including concerted transglycosylation. Apparent energetics were compared with computed literature values.
- This article is part of the themed collection: Reaction Chemistry & Engineering Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...