Catalytic synergy effect of MoS2/reduced graphene oxide hybrids for a highly efficient hydrogen evolution reaction†
Abstract
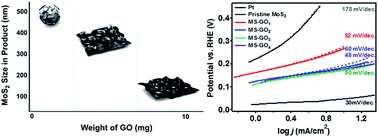
Two-dimensional layered transition metal dichalcogenide (TMD) materials such as MoS2 and WS2 have received a great deal of attention as alternatives to Pt in hydrogen evolution reaction (HER) catalysts. Recently, confined synthesis of TMD nanoparticles on graphene exhibited great catalytic performance for HER, due to the presence of many active edge sites. However, the correlation of gradual electronic transition states of TMD/graphene hybrids to catalytic behavior has been rarely studied. By means of controlling only the graphene oxide (GO) content added during the solvothermal synthesis with MoS2, we have synthesized hybrids of MoS2 and reduced graphene oxide (rGO) with tunable morphology; this tuning also brought about a gradual change in the electronic states of MoS2 due to strain induced by van der Waals interaction between heterolayers. The GO content tuning gradually enhanced the HER catalytic performance of the MoS2/rGO hybrids, decreasing the Tafel slope from 82 to 48 mV per decade owing to synergistic effects of an increase of catalytically active areas, an electronic transition of MoS2, and conductivity of rGO substrates.


 Please wait while we load your content...
Please wait while we load your content...