Size-selective adsorption of anionic dyes induced by the layer space in layered double hydroxide hollow microspheres†
Abstract
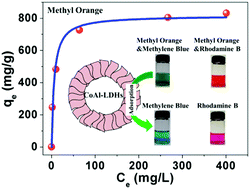
Flower-like cobalt aluminum layered double hydroxide (CoAl-LDH) hollow microspheres were synthesized via a one-step solvothermal method without any template. An Ostwald ripening mechanism was proposed for the formation of hollow nanostructures. These flower-like CoAl-LDH hollow microspheres had a high surface area and exhibited excellent selectivity for anionic dyes. The limited space between LDH layers offered size selectivity for adsorbate molecules. For the small molecule methyl orange, the maximum adsorption capacity reached 816.0 mg g−1 under ambient conditions, while for larger molecules such as Eosin B, the adsorption capacity was only 95.1 mg g−1. All these features make the flower-like CoAl-LDH hollow microspheres an excellent adsorbent in water remediation.


 Please wait while we load your content...
Please wait while we load your content...