Aromatic-rich hydrocarbon porous networks through alkyne metathesis†
Abstract
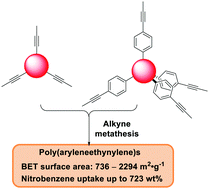
Purely hydrocarbon-based porous polymers have generally been prepared through various irreversible transition metal-catalyzed cross-coupling reactions forming C–C bonds. Herein, we report an alternative synthetic approach, namely reversible alkyne metathesis, for the preparation of ethynylene-linked porous polymers. Planar and tetrahedral-shaped monomers were explored to construct poly(aryleneethynylene) (PAE) networks. We systematically varied the size of the monomers and studied the structure–property relationships. The resulting polymers exhibit high Brunauer–Emmett–Teller (BET) surface areas in the range of 736 m2 g−1 to 2294 m2 g−1. The advantages of such aromatic-rich PAE networks are their lightweight, high thermal/chemical stabilities, and superior hydrophobicity, which are beneficial for their application in adsorption/separation of toxic organic pollutants from water. We found that PAEs can adsorb a significant amount of common aromatic solvents, e.g. up to 723 wt% of nitrobenzene. Our study thus demonstrates an encouraging novel approach to prepare purely hydrocarbon-based porous materials.
- This article is part of the themed collection: Functional Open framework materials


 Please wait while we load your content...
Please wait while we load your content...