Improvement of D–π–A organic dye-based dye-sensitized solar cell performance by simple triphenylamine donor substitutions on the π-linker of the dye†
Abstract
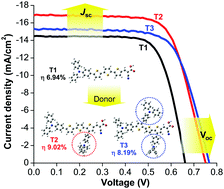
We report a new simple way to improve the performance of a basic D–π–A organic dye in dye-sensitized solar cells (DSSCs) by donor substitutions on the π-linker of the dye. Three new molecularly engineered D–π–A dyes, namely T2-4, comprising triphenylamine (TPA) as a donor, terthiophene containing different numbers of TPA substitutions as a π-conjugated linker and cyanoacrylic acid as an acceptor, were synthesized and characterized. A detailed study on the effect of different dye structures on the performance of the DSSCs was conducted systematically using theoretical, photophysical, photovoltaic as well as photoelectrochemical methods and compared with that of the traditional D–π–A dye, namely T1. The introduction of electron donating TPA substitutes on the π-linker of the D–π–A dye were beneficial for a decrease of the electron recombination between redox electrolyte and the TiO2 surface as well as an increase of the electron correction efficiency, leading to improved open-circuit voltage (VOC) and short-circuit current (JSC). Consequently, DSSCs sensitized by dye T2 bearing one extra TPA substitution on the terminal thiophene ring of the π-linker delivered the best power conversion efficiency, reaching 8.08% at AM 1.5 simulated sunlight, a remarkable improvement of about 41% compared with 5.72% of T1 reference cells, owing to its high JSC and VOC. With the addition of CDCA as a coadsorbent, the best performing cells based on dye T2 exhibited an impressive conversion efficiency of 9.02% (JSC = 16.91 mA cm−2, VOC = 754 mV, FF = 0.705), exceeding that of the N719-based standard cell (8.20%). This renders the facile dye modification very promising to acquire simple effective organic dyes for high-efficiency DSSCs.


 Please wait while we load your content...
Please wait while we load your content...