A novel radiation chemistry-based methodology for the synthesis of PEDOT/Ag nanocomposites
Abstract
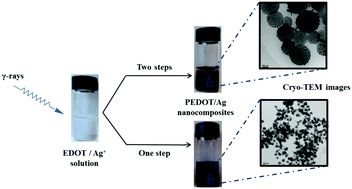
An original methodology based on radiation chemistry was used for the synthesis of hybrid organic–inorganic composites in aqueous solution. Starting from 3,4-ethylenedioxythiophene (EDOT) monomers and silver perchlorate salt, the preparation of PEDOT/Ag composites, made of poly(3,4-ethylenedioxythiophene) (PEDOT) conducting polymers and silver nanoparticles, was achieved through γ-radiolysis via different procedures. According to a two-step method, PEDOT polymers were first synthesized by reduction or oxidation of the EDOT monomers, and then silver nanoparticles were produced in the presence of PEDOT by the reduction of silver ions. According to a one-pot method, PEDOT polymers and silver nanoparticles were synthesized simultaneously thanks to the concomitant reduction of EDOT monomers and silver ions, as demonstrated via pulse radiolysis experiments. As highlighted using UV-Vis absorption spectrophotometry, ATR-FTIR spectroscopy and EDX spectroscopy, all the prepared PEDOT/Ag nanocomposites have the same chemical composition and are found to be doped, after lyophilization, with perchlorate anions. Nevertheless, due to the different synthesis procedures, the morphologies of the nanocomposites are found to be almost different as demonstrated using cryo-TEM and SEM microscopies. TGA revealed that, whatever their morphology, all the PEDOT/Ag composites are characterized by the same thermal stability, which is higher than that of pure PEDOT and which is enhanced upon increasing the amount of silver. Finally, four-point probe measurements demonstrated that the values of electrical conductivity of all the radiosynthesized PEDOT/Ag composites are in the same order of magnitude and remain close to those reported in the literature for PEDOT nanocomposites prepared according to other methodologies.

 Please wait while we load your content...
Please wait while we load your content...