Controlled release of antibody proteins from liquid crystalline hydrogels composed of genetically engineered filamentous viruses†
Abstract
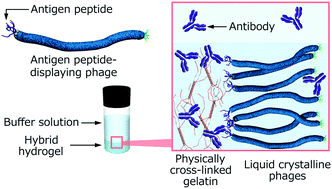
Affinity-based controlled-release systems have attracted attention for drug delivery owing to the avoidance of burst or fast release and because they provide tunable release properties. To widen the applicability of the system, construction of universal matrices, which enable the control of drug molecule release, is essential. Herein, we constructed controlled-release systems for high molecular weight antibody proteins using genetically engineered filamentous viruses, M13 phages. Hybrid hydrogels composed of antigen HA peptide-displaying phages (HA-phages) and physically cross-linked gelatin containing anti-HA peptide antibodies (HA-antibodies) were prepared for controlled-release systems. Quantification of the antibody molecules released from the hybrid hydrogels revealed that specific interactions between the phage-displayed HA peptides and antibodies were necessary for suppression of the release. The importance of the liquid crystalline-ordered lamellar structures of the assembled phages for effective suppression was evidenced by polarized optical microscopy and quantification of dialyzed amounts of HA-antibodies. The release rates and amounts could be controlled by altering the concentration of HA-phages (i.e., the concentration of the displayed HA peptides). Furthermore, utilization of the combination of HA-phages and another antigen, FLAG peptide-displaying phages, in the hybrid hydrogel resulted in the precise, controlled release of multiple antibodies from a single hybrid hydrogel. The use of genetically engineered phages as hydrogel matrices creates attractive opportunities for the science and technology of hydrogels with controlled release.

 Please wait while we load your content...
Please wait while we load your content...