Improving fiber alignment by increasing the planar conformation of isoindigo-based conjugated polymers
Abstract
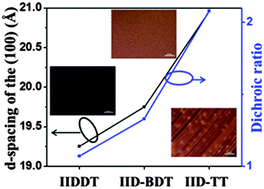
The macroscopic alignment of conjugated polymers with few grain boundaries is essential for carrier transport. It is well known that molecular conformation has a big role in achieving macroscopic alignment. Therefore, in this paper, a series of isoindigo-based conjugated polymers, the copolymer of isoindigo and thieno-[3,2-b]thiophene (IID–TT), isoindigo and bithiophene (IIDDT) and isoindigo and benzodithiophene (IID–BDT), were chosen to investigate the effect of molecular conformation on the fiber alignment. The long-range ordered film was obtained for IID–TT by drop-casting and solvent vapor enhanced drop-casting (SVE DC). The dichroic ratio (DR) of the IID–TT film obtained by SVE DC was 2.08 higher than that of the other two isoindigo-based conjugated polymers (IIDDT and IID–BDT), 1.07 and 1.33, respectively. The fibrils in the IID–TT film obtained by SVE DC were parallel and grown along the molecular backbones, but the fibrils of IIDDT and IID–BDT were both isotropic. The torsion angle of IID–TT (isoindigo and thieno-[3,2-b]thiophene) is 1.22°, while the torsion angle of IIDDT (isoindigo and thiophene, thiophene and thiophene) is 0.97° and 0.38° and that of IID–BDT (isoindigo and benzodithiophene) is 1.32°, respectively. The molecular conformation of IID–TT is the most coplanar among the three copolymers. The alkyl-stacking distance of IID–TT was 20.96 Å larger than that of IIDDT (19.25 Å) and IID–BDT (19.75 Å) which indicated that the interaction of the side-chain of IID–TT was the weakest among the three conjugated polymers. The DR increased with the increase in the alkyl-stacking distance. The weak interaction of the side-chain in IID–TT was beneficial for the rearrangement of molecules in parallel. The coplanar conformation could accelerate interchain assembly of the parallel molecules through π–π interaction to form parallel fibers.

 Please wait while we load your content...
Please wait while we load your content...