Palladium complexes of ferrocene-based phosphine ligands as redox-switchable catalysts in Buchwald–Hartwig cross-coupling reactions†
Abstract
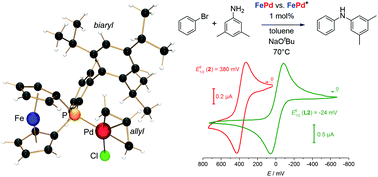
The synthesis and full characterization of several transition metal complexes of a redox-switchable, biaryl(ArT)-substituted [1]phosphaferrocenophane, FcPArT (L2), in conjunction with some palladium complexes of the mesityl(Mes)-substituted derivative, FcPMes (L1), and the non-bridged diphenylferrocenyl phosphine, Ph2PFc (L3), are reported. Cyclic voltammetry studies on the bimetallic complexes [Ir(κ1P-L2)(cod)Cl] (1), [Ir(κ1P-L2)(CO)2Cl] (2), [AuCl(κ1P-L2)] (3), [Pd(κ1P-L2)(η3-allyl)Cl] (4), [Pd(κ1P-L3)(η3-allyl)Cl] (5), [Pd(κ1P-L3)(2-aminobiphenyl)(SO3CH3)] (6), and [Pd(κ1P-L1)(2-aminobiphenyl)(SO3CH3)] (7) provided indications for a good electronic communication between the metal atoms. In order to confirm that the [1]ferrocenophane or 1-ferrocenyl unit might be able to electrochemically influence the reactivity of the coordinated transition metal, the palladium complexes 4–7 were employed as redox-switchable catalysts (RSC) in Buchwald–Hartwig cross-coupling reactions. The catalytic activity depends on the ligand and decreases in the series L3 > L2 > L1. In stirred solutions, the reaction rate can be influenced by addition of an oxidizing reagent. Some reactions were accelerated with in situ generated, cationic complexes as catalysts as compared to their neutral analogs; in some cases, the activity was found to be lower. Furthermore, steric effects play a predominant role, among other factors.
- This article is part of the themed collection: Switchable Catalysis and Related Reactions


 Please wait while we load your content...
Please wait while we load your content...