Mechanistic study of graphitic carbon layer and nanosphere formation on the surface of T-ZnO†
Abstract
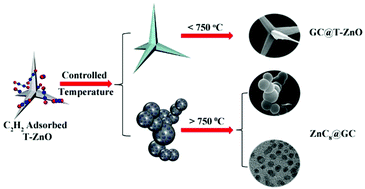
Graphitic carbon (GC) coatings on the surface of functional materials have received tremendous attention because of their significant effect on surface chemistry. Herein, we have explained the interactions of a carbon source (acetylene) on the surface of functional material (tetrapod-zinc oxide, T-ZnO) through Density Functional Theory (DFT) calculations. DFT studies suggested that the growth of GC in the catalytic chemical vapour deposition (CCVD) process follows two typical pathways: one is chemo/physioadsorption of acetelyne on the T-ZnO surface, and the second one is decomposition of acetylene on the T-ZnO surface to activate carbon atoms that are restructured to generate a GC layer on the T-ZnO surface. Therefore, such a growth through catalytic accumulation reaction of carbon species on the substrate surface is named as vapour-dissociation-solid (VDS) growth. To support our theoretical observation, the GCs on the surface of T-ZnO are grown using acetylene through CCVD, and the results strongly support the VDS growth mechanism. The varying conditions of the CCVD process results in two types of microstructures including a smooth GC coating on T-ZnO that can also yield functional hollow GC nanostructures through acid leaching of the metallic part and ZnC8 nanoparticles embedded in GC walls. The high resolution microscopic analyses confirm the existence of ZnC8 in the nanospheres with an average size of 5.36 nm, which authenticates the decomposition followed by the solid deposition to form the GC on the substrate surface and supports a unique VDS growth mechanism. Therefore, an in-depth study of the GC growth mechanism and its fine coating on complex structures will facilitate research in the prevention of metal oxidation and the design of novel active materials for sensors, environment, energy and medical applications.

 Please wait while we load your content...
Please wait while we load your content...