A comparative study of the effect of functional groups on C2H2 adsorption in NbO-type metal–organic frameworks†
Abstract
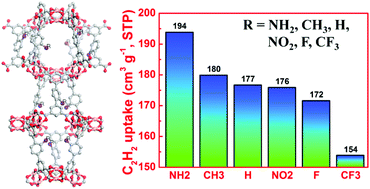
Investigation of the effect of functional groups on gas adsorption in a MOF of a given structure is undoubtedly very important because it facilitates targeting porous MOFs with enhanced storage capacities by ligand functionalization. In this work, we chose an NbO-type MOF platform to evaluate the impact of organic functional groups on C2H2 adsorption. Correspondingly, we synthesized five diisophthalate ligands with the same length but different organic functionalities (NH2, CH3, NO2, F, and CF3), and successfully incorporated them into MOFs with underlying NbO topology. C2H2 adsorption experiments reveal that these functional moieties can enhance the affinity towards C2H2, but not all these compounds outperform the unfunctionalized parent compound NOTT-101 in terms of C2H2 uptake. At 298 K and 1 atm, the C2H2 uptake varies from 153.7 to 193.8 cm3 (STP) g−1, depending on the attached organic functional groups. Particularly, the amine group-functionalized compound ZJNU-34(NH2) exhibits the maximum C2H2 uptake of the six compounds evaluated, reaching as high as 203.6 cm3 (STP) g−1 at 295 K and 1 atm. Such a 10% increase of C2H2 uptake compared to the parent compound might be attributed to acid–base and/or hydrogen-bonding interactions between the NH2 groups with adsorbed C2H2 molecules. The fundamental understanding of the impact of functional groups on C2H2 adsorption demonstrated in this work provides valuable information for future designing of porous MOFs with enhanced acetylene-storage capacities.


 Please wait while we load your content...
Please wait while we load your content...