Synthesis, crystal and electronic structures, and physical properties of a new quaternary phosphide Ba4Mg2+δCu12−δP10 (0 < δ < 2)†‡
Abstract
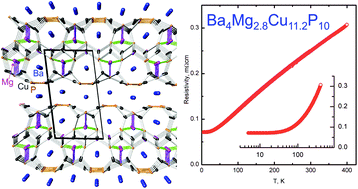
A new quaternary phosphide, Ba4Mg2+δCu12−δP10 (0 < δ < 2), was synthesized via solid state reactions of the elements and its crystal structure was determined by single crystal X-ray diffraction. Ba4Mg2.8Cu11.2P10 crystallizes in the monoclinic system space group P21/m (no. 11) with unit cell parameters of a = 8.258(1) Å, b = 4.009(1) Å, c = 15.260(2) Å, β = 97.851(2)°, and Z = 1. The crystal structure of Ba4Mg2+δCu12−δP10 is composed of large two-dimensional Ba2Mg2+δCu12−δP10 blocks stuck along the [001] direction. Each block is terminated by unique Cu4P3 layers with a PbO-like MgδCu2−δP2 layer inside the block. Cu4P3 and MgδCu2−δP2 layers are linked together in the block by Cu–P bonds. All Mg and half of the Ba cations are situated in the large channels inside the blocks while the other half of Ba atoms are arranged in corrugated layers separating the blocks. The metallic properties of Ba4Mg2.8Cu11.2P10 were predicted based on electronic structure calculations and confirmed by resistivity measurements. Ba4Mg2.8Cu11.2P10 exhibits moderate thermal conductivity, 13.4 W m−1 K−1 at 400 K, which mainly originates from the lattice contribution of 9.1 W m−1 K−1 at 400 K.
- This article is part of the themed collection: In honour of Mercouri G. Kanatzidis for his contributions to Inorganic Chemistry for over 30 years


 Please wait while we load your content...
Please wait while we load your content...