S,N-Containing Co-MOF derived Co9S8@S,N-doped carbon materials as efficient oxygen electrocatalysts and supercapacitor electrode materials†
Abstract
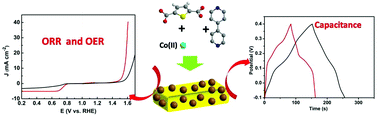
Controllable synthesis of metal–organic framework (MOF) materials with tunable morphologies, sizes, compositions and pore structures is critically important for MOF materials and their pyrolysis derivatives’ applications in environmental and energy fields. Here we report the synthesis of Co-MOF crystals with controllable morphologies, sizes and S/N ratios in water/NaOH and ethylene glycol/NaOH systems using thiophene-2,5-dicarboxylate (Tdc) and 4,4′-bipyridine (Bpy) as S, N dual organic ligands by a “pillar-layer” assembly method. Water and ethylene glycol with different viscosities result in various crystallization processes of [Co(Tdc)(Bpy)]n crystals in the corresponding reaction system, thus respectively obtaining three-dimensional (3D) Co-MOF ([Co(Tdc)(Bpy)]n) bulk and cuboid structures in water/NaOH and ethylene glycol/NaOH reaction systems. The as-prepared Co-MOF crystals in two different reaction systems were further pyrolytically treated at 800 °C in a N2 atmosphere to obtain Co9S8@S,N-doped carbon materials with different surface areas, pore distributions and S/N doping ratios. As electrocatalysts, the Co9S8@S,N-doped carbon cuboid (Co9S8@SNCC) obtained in the ethylene glycol/NaOH system demonstrates superior bifunctional electrocatalytic activities toward both oxygen reduction and evolution reactions resulting from Co9S8 and S, N doping in the carbon structure providing catalytic active sites, better than that of Co9S8@S,N-doped carbon bulk (Co9S8@SNCB) obtained in the water/NaOH system and comparable to commercial Pt/C and RuO2 catalysts. Owing to its high surface area and porous structure, Co9S8@SNCC also exhibits great potential as the electrode material for application in supercapacitors, with high performance and recycling stability.
- This article is part of the themed collection: Inorganic Chemistry Frontiers HOT articles for 2017

 Please wait while we load your content...
Please wait while we load your content...