Synthesis and sequence-controlled self-assembly of amphiphilic triblock copolymers based on functional poly(ethylene glycol)†
Abstract
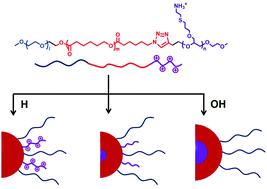
Given the increasing prosperity of multifunctional poly(ethylene glycol) (mf-PEG), an amphiphilic triblock copolymer, poly(ethylene glycol)-block-poly(ε-caprolactone)-block-poly(allyl glycidyl ether) (mPEG-PCL-PAGE), was synthesized by a combination of living ring-opening polymerization (ROP) and click chemistry. A post-modification of the allyl groups can be achieved via a radical mediated thiol–ene reaction to prepare copolymers bearing side-chain carboxylic or amino groups [mPEG-PCL-PAGE(R)]. The chemical structures and compositions of these polymers were systematically characterized using Fourier transform infrared spectroscopy (FT-IR), proton nuclear magnetic resonance spectroscopy (1H NMR) and gel permeation chromatography (GPC). The copolymers could readily assemble into micelles in aqueous solution as confirmed by fluorescence spectroscopy, dynamic light scattering (DLS), transmission electron microscopy (TEM) and zeta potential measurements. MTT and hemocompatibility assays indicated that the micelles showed no associated cytotoxicity and could be potentially used for nanomedicine. Intriguingly, the specific sequence of [mPEG-PCL-PAGE(R)] showed pH-induced morphology transformation, while the corresponding polymer with a middle PAGE segment [mPEG-PAGE(R)-PCL] did not, which, we propose, was attributed to the tunable hydrophilicity and distinctive folding of the molecular chain at varied pH values. This finding offers a route to regulate the self-assembly behaviors in polymer systems by controlling the block sequence.


 Please wait while we load your content...
Please wait while we load your content...