Post-synthetic modification of polyvinyl alcohol with a series of N-alkyl-substituted carbamates towards thermo and CO2-responsive polymers†
Abstract
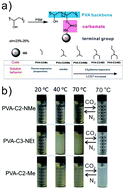
Intensive efforts have been devoted to the synthesis of thermoresponsive polymers with terminal N-alkyl-substituted groups. Furthermore, an investigation into polymers with a lower critical solution temperature (LCST) controlled by CO2 tuning is important to many applications; yet, this technology cannot advance without understanding the critical impact that N-alkyl-substituted groups has on the polymer. These polymers have been synthesized by polymerization of (co)monomers (e.g., acrylamide or methacrylate) with N-alkyl-substituted groups in a block or random configuration. Complicated synthesis procedures and unconfirmed biocompatibility may limit their desired properties. In this work, a general post-synthetic modification strategy is developed to functionalize poly(vinyl alcohol) (PVA) with a series of pendant alkyl or N-alkyl-substituted carbamates under facile conditions. The resulting PVA derivatives show excellent biocompatibility. The mechanism study shows that the LCST behavior of the PVA derivatives with pendant N-alkyl-substituted carbamates is attributed to the hydrogen bonding effect and hydrophobic interactions with terminal tertiary amines. However, a PVA derivative without terminal tertiary amines will likely exhibit globular conformation in solution, even at low temperatures (e.g. 15 °C). Heating induces slow agglomeration, but no changes in the hydrogen bonding occur. In addition to the conformation changes, heating can induce fluctuations in the pKa of tertiary amines contained in PVA derivatives, which facilitates the CO2 absorption/desorption cycles. Below the phase transition temperature, most amines in PVA derivative solutions are capable of being protonated in the presence of CO2, resulting in a significant increase in the LCST. Above the phase transition temperature (70 °C), the hydrophilic-to-hydrophobic transition of PVA derivatives lowers the pKa, thereby resulting in an almost complete desorption of CO2 upon N2 bubbling. This work demonstrates a general post-synthetic modification in the synthesis of responsive polymers, the significance of the alkyl amine motif in the solution behavior and the potential of thermoresponsive polymer solutions to capture CO2.


 Please wait while we load your content...
Please wait while we load your content...