Synthesis of molecular brushes by telomerization†
Abstract
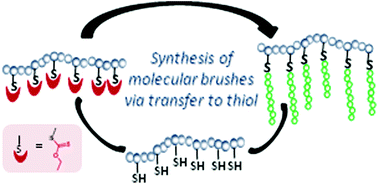
A synthetic methodology to prepare molecular brushes via radical transfer is presented. This method relies on the use of polythiol copolymers as multifunctional macrotransfer agents to grow side-chains via radical transfer to pendant thiols. To test this methodology, polymethacrylate copolymers with ca. 25 and 50 mol% of repeating units carrying a pendant protected thiol were prepared by radical polymerization, using an alkyl xanthate as the thiol protecting group. One-pot two-step and two-pot reaction sequences were tested for thiol deprotection and side-chain growth. Using transfer to pendant thiols, molecular brushes with side-chains of various natures, e.g. methacrylate, acrylate and acrylamide, and the degree of polymerization, ranging from 20 to 70, were prepared. Kinetic studies indicated that side-chains grown at different stages of the polymerization could display significantly different degrees of polymerization, which reflects both the drift of the [M]/[SH] ratio and the change of the transfer constant during the polymerization. By using the appropriate ratio of the radical initiator to thiol groups, it was demonstrated that brushes with no detectable free chains could be prepared. The synthetic methodology described in this study offers a straightforward and robust route to prepare molecular brushes with an adjustable grafting density, side-chain nature and degree of polymerization.
- This article is part of the themed collection: Pioneering Investigators


 Please wait while we load your content...
Please wait while we load your content...