Effect of conjugation length and metal-backbone interactions on charge transport properties of conducting metallopolymers†
Abstract
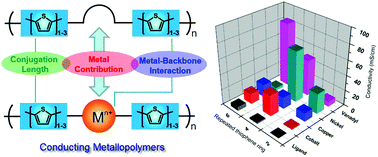
A systematic study on the effect of variation in length of the conjugated organic backbone, and therefore the redox interactions between metal centers and the polymer backbones, of thiophene-based conducting metallopolymers (CMPs) on their charge transport properties has been conducted. Schiff-base ligands bearing various numbers of thiophene units (1–3) on both sides of a salpen center (salicyaldehyde connected through a 1,3-propylenediamine backbone) and the corresponding metal complexes of both redox-active and redox-inactive metals have been synthesized and characterized. Conducting polymers from these electropolymerizable monomers were obtained via electrochemical synthesis and studied by cyclic voltammetry, vis-NIR spectroelectrochemistry, and in situ conductivity. The results indicate that charge transport properties of ligand polymers are dramatically affected by the conjugation length of the organic backbone. The conductivity of ligand polymers increases by approximately one order of magnitude when varying the repeating units of the polymers from a bithiophene (2T) to a quarterthiophene (4T) or from a 4T to a sexithiophene (6T). However, the conductivity of the metallopolymers is significantly influenced by the interactions between the metal centers and the organic backbone. Copper(II) metallopolymers, which show no metal redox event, exhibit similar conductivity for CMPs bearing different conjugated lengths of the organic backbone. CMPs of redox-active metals (i.e., Ni(II), Co(II), and V(IV)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), on the other hand, show significant changes in conductivity depending on the relative redox potential of the metal centers and that of the organic backbone. Among metallopolymers which bear the same metal, CMPs with redox matching exhibit the highest conductivities in the series. We define redox matching as the condition in which the redox potential of the metal center is slightly higher than, but does not exceed, that of the organic backbone by ca. 0.5 V. The studies herein provide insights in understanding the structure–property relationship of CMPs, particularly on the role of the organic conjugated length, the metal centers, and metal-backbone interactions on the charge transport properties of Wolf Type III CMPs.
O), on the other hand, show significant changes in conductivity depending on the relative redox potential of the metal centers and that of the organic backbone. Among metallopolymers which bear the same metal, CMPs with redox matching exhibit the highest conductivities in the series. We define redox matching as the condition in which the redox potential of the metal center is slightly higher than, but does not exceed, that of the organic backbone by ca. 0.5 V. The studies herein provide insights in understanding the structure–property relationship of CMPs, particularly on the role of the organic conjugated length, the metal centers, and metal-backbone interactions on the charge transport properties of Wolf Type III CMPs.


 Please wait while we load your content...
Please wait while we load your content...