Switchable 19F MRI polymer theranostics: towards in situ quantifiable drug release†
Abstract
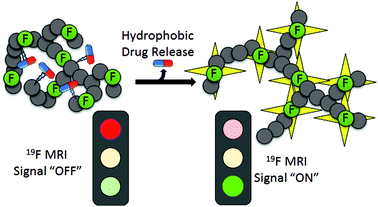
A switchable polymeric 19F magnetic resonance imaging (MRI) contrast agent was synthesised whereby the transverse (T2) relaxation times increased as a therapeutic was released from a hyperbranched polymer (HBP) scaffold. The HBP comprised of poly(ethyleneglycol) monomethyl ether methacrylate (PEGMA), a fluorinated monomer (trifluoroethyl acrylate), and a suitable monomer for post-conjugation of a drug molecule. Three different hydrophobic drugs were investigated during design of the theranostic; doxorubicin (DOX) and docetaxel (DTX) were conjugated to the HBPs through an acid-cleavable hydrazone linkage, while camptothecin (CPT) was integrated into the HBP via polymerisation of a self-immolative disulphide-linked monomer. 19F NMR relaxometry measurements showed that the increase in hydrophobicity caused by the incorporation of the therapeutic drug led to a decrease in 19F T2 relaxation times and decrease in image intensity. However, upon drug release, the hydrophobicity of the HBP decreased which in turn led to improved mobility of the fluorinated moieities. This was manifest in a restoration of longer 19F T2 relaxation times and an increased image intensity compared to the drug-loaded polymer. This work provides a basis for a MRI contrast agent capable of quantifying in situ drug release.
- This article is part of the themed collection: Pioneering Investigators


 Please wait while we load your content...
Please wait while we load your content...