One-pot synthesis of pH-responsive hyperbranched polymer–peptide conjugates with enhanced stability and loading efficiency for combined cancer therapy†
Abstract
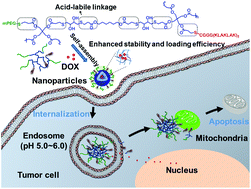
Nanoparticles as drug-delivery systems have received significant attention due to their merits such as prolonged circulation time and passive targeting of a tumor site. Polymer–peptide conjugates (PPCs) tend to self-assemble into nanoparticles in an aqueous solution, and the resulting nanoparticles as drug carriers combine the virtues of both the polymers and peptides. In this study, a simple synthetic method based on a thiol–acrylate Michael addition reaction was used for the one-pot synthesis of amphiphilic hyperbranched poly(β-thioester)s (PPHD-PK) conjugated with cytotoxic peptide (KLAKLAK)2 (denoted as KLAK) and poly(ethylene glycol) (PEG). In aqueous media, PPHD-PK self-assembled into nanoparticles, and the hyperbranched poly(β-thioester)s (PPHD) containing acid-labile β-thiopropionate group acted as the interior of the nanoparticles, whereas PEG and KLAK were employed as the outer shell. The PPHD-PK nanoparticles showed enhanced cellular uptake and favorable antitumor activity, which was attributed to the spherical structure with superficial positive charges and mitochondria-regulated apoptosis of the KLAK peptide. Compared with linear PPCs, the stability of the nanoparticles and the drug-loading efficiency of PPHD-PK were significantly improved, implying that a stronger intermolecular interaction was generated by intertwisting of the branched networks in the nanoparticle core region. Doxorubicin (DOX), as a typical chemotherapeutic drug, was readily released from PPHD-PK under the acidic environment of lysosome, thus leading to efficient nuclear drug translocation and resultant potent drug efficacy. Furthermore, DOX-loaded PPHD-PK nanoparticles showed higher cytotoxic activity than DOX-loaded PPHD-P (without KLAK) and blank PPHD-PK nanoparticles, indicating that the combined treatment of DOX and KLAK was most effective to kill HeLa cells. Therefore, DOX-loaded PPHD-PK nanoparticles with enhanced stability and loading efficiency exhibit great potential as antitumor nanodrugs for efficient cancer therapy.


 Please wait while we load your content...
Please wait while we load your content...