Retracted Article: Ni/Co/Ti layered double hydroxide for highly efficient photocatalytic degradation of Rhodamine B and Acid Red G: a comparative study†
Abstract
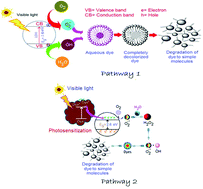
Optically responsive, luminescent Ni/Co/Ti layered double hydroxide (LDH), synthesized by a single step hydrothermal route, exhibits highly efficient photodegradation of cationic and anionic dyes, Rhodamine B (∼99.8%) and Acid Red G (∼99.6%) respectively, better than that of commercial catalysts like NiO, CoO and TiO2. The LDH has been characterized by using XRD, XPS, PL, TRES, EIS, TEM, SEM-EDX, AFM, UV-visible DRS, N2-sorption desorption, ξ-potential, FT-IR and TG techniques. The characterized results indicate that the LDH possesses hexagonal morphology, a high surface area, a narrow band gap, defect states and oxygen vacancies within its layered framework. The degradations follow the e−–h+ hopping pattern and dye-photosensitized mechanistic pathways. The active species generated during photocatalysis have been evaluated using ESR, terephthalic acid fluorescence probe and indirect radical–hole trapping experiments. The colourless end products were investigated by GC-MS and reaction mechanisms have been established for the degradation of the dyes to less toxic and more eco-friendly molecules than their parent analogues. Dye mineralization studies (performed using a TOC analyser) and closure of carbon mass balance experiments quantified the amount of carbon entering and leaving the reaction systems. Reaction mechanisms have been proposed on the basis of the asymmetric cleavage of the dyes. The LDH demonstrated its remarkable efficiency in the field of waste water treatment.


 Please wait while we load your content...
Please wait while we load your content...