Effect of the calcination temperature on the photocatalytic efficiency of acidic sol–gel synthesized TiO2 nanoparticles in the degradation of alprazolam†
Abstract
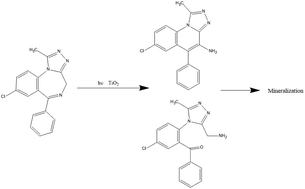
We report a comparative study on the photodegradation of the widely used benzodiazepine psychoactive drug alprazolam (8-chloro-1-methyl-6-phenyl-4H-[1,2,4]triazolo[4,3-a][1,4]benzodiazepine, ALP) using direct photolysis, and titanium dioxide photocatalyzed reaction. Titanium dioxide photocatalysts were prepared as nanoparticles by acidic sol–gel methods, calcined at two different temperatures, and their behavior compared with P25 (Degussa type) TiO2. Efficient photodegradation was observed in the photocatalytic process, with over 90% degradation after 90 minutes under optimized conditions. Triazolaminoquinoline, 5-chloro-(5-methyl-4H-1,2,4-triazol-4-yl)benzophenone, triazolbenzophenone, and α-hydroxyalprazolam were identified as the degradation products by fluorescence spectroscopy and HPLC-MS. A comparison with the literature suggests that 8H-alprazolam may also be formed. Good mineralization was observed with TiO2 photocatalysts. ALP photodegradation with TiO2 follows pseudo-first order kinetics, with rates depending on the photocatalyst used. The effects of the quantity of the photocatalyst and concentration of alprazolam were studied.


 Please wait while we load your content...
Please wait while we load your content...