Effect of anthraquinone-2,6-disulfonate on the photolysis of 2,4,4′-tribromophenylphenyl ether
Abstract
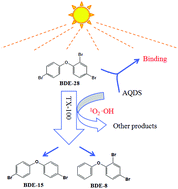
In this study, we have investigated the photolysis of 2,4,4′-tribromophenylphenyl ether (BDE-28) in Triton X-100 (TX-100) solutions, and discussed the effect of anthraquinone-2,6-disulfonate (AQDS) on the photolysis of BDE-28. The effect of TX-100 on the photolysis of BDE-28 was mainly related to the concentration of TX-100. The fastest photolysis of BDE-28 was at 500 mg L−1 of the TX-100 solution, and the corresponding photolysis rate constant was 0.12 min−1. The direct photolysis rate of BDE-28 decreased from 0.17 min−1 to 0.08 min−1 and 0.12 min−1 when NaN3 and isopropanol were added, respectively. The effect of AQDS on the photolysis of BDE-28 was also mainly related to the concentration of AQDS. When the concentration of AQDS was 0.6 μM, it has a slight influence in promoting the photodegradation of BDE-28; as the AQDS concentrations increased, the suppressing effect was more obvious. AQDS can inhibit the photolysis of BDE-28. Photolysis kinetics and quenching reactions illustrated that BDE-28 can produce a photosensitization reaction in TX-100 solutions, and the effects of AQDS on the photolysis of BDE-28 were mainly dominated by inhibition. In addition to its light shielding effect, AQDS can also combine with BDE-28 and anti-oxidation to inhibit the photolysis of BDE-28. We found that the degradation pathway of BDE-28 was mainly based on de-bromination, and the ions at para positions were preferentially debrominated.


 Please wait while we load your content...
Please wait while we load your content...