Effect of nucleants in photothermally assisted crystallization†
Abstract
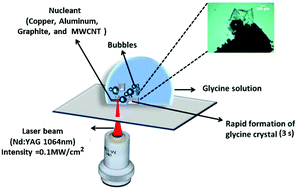
Laser-induced crystallization is emerging as a promising technique to crystallize biomolecules like amino acids and proteins. The use of external materials as nucleants and novel seeding methods open new paths for protein crystallization. We report here the results of experiments that explore the effect of nucleants on laser-based crystallization of microlitre droplets of small molecules, amino acids, and proteins. The role of parameters like solute concentration, droplet volume, type and size of the nucleant, and laser power, are systematically investigated. In addition to crystallization of standard molecules like NaCl, KCl, and glycine, we demonstrate the crystallization of negatively (L-histidine), and positively (L-aspartic acid) charged amino acids and lysozyme protein. Single crystal X-ray diffraction and Raman spectroscopy studies unequivocally indicate that the nucleants do not alter the molecular structure of glycine, hydrogen bonding patterns, and packing. Localized vaporization of the solvent near the nucleant due to photothermal heating has enabled us to achieve rapid crystallization – within 3 s – at laser intensities of 0.1 MW cm−2, significantly lower than those reported earlier, with both saturated and unsaturated solutions. The outcome of the current experiments may be of utility in tackling various crystallization problems during the formation of crystals large enough to perform X-ray crystallography.


 Please wait while we load your content...
Please wait while we load your content...