A spectroscopic study of the excited state proton transfer processes of (8-bromo-7-hydroxyquinolin-2-yl)methyl-protected phenol in aqueous solutions†
Abstract
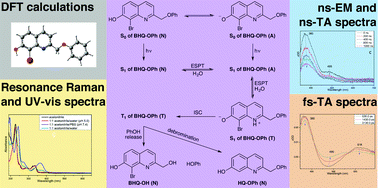
A combination of spectroscopic methods and density functional theory (DFT) computations was used to study the excited state proton transfer (ESPT) processes of (8-bromo-7-hydroxyquinolin-2-yl)methyl-protected phenol (BHQ-OPh). Characterization of the prototropic forms of BHQ-OPh in different solvent environments revealed that the neutral form predominates in acetonitrile and in 1 : 1 acetonitrile/water (pH 5.0), whereas the anionic form predominates in 1 : 1 acetonitrile/PBS (pH 7.4). Both the neutral and anionic forms were significantly populated in 1 : 1 acetonitrile/water. Upon irradiation in acetonitrile the triplet neutral form was observed, whereas the triplet anionic form was detected in 1 : 1 acetonitrile/PBS (pH 7.4). The existence of the triplet tautomeric form of BHQ-OPh in both 1 : 1 acetonitrile/water and 1 : 1 acetonitrile/water (pH 5.0), and the ESPT processes from the neutral to the anionic to the tautomeric forms in the excited state were observed using time-resolved spectroscopy. A reaction mechanism in 1 : 1 acetonitrile/water and 1 : 1 acetonitrile/water (pH 5.0) was proposed based on the spectroscopic and DFT computational results. A comparison of the results for BHQ-OPh with those of BHQ-OAc reveals that the initial prototropic states and photochemical processes are similar. The understanding gained of the initial photo-induced processes of BHQ-based photoremovable protecting groups (PPGs) is useful for the design of new quinolinyl-based PPGs for specialized applications.


 Please wait while we load your content...
Please wait while we load your content...