Photo-induced phosphate released from organic phosphorus degradation in deionized and natural water†
Abstract
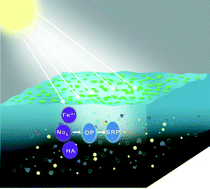
The photodegradation of organic phosphorus is one of the most important processes of the phosphorus cycle by which phosphate is regenerated in the water environment. In this study, the influence of direct photolysis or indirect photolysis of organic phosphorus using natural photosensitizers on the released phosphate was examined in deionized and natural water under ultraviolet (UV) irradiation using diazinon as the organic phosphorus model. Phosphate was released when diazinon was exposed to UV light, and the solution pH also exhibited distinct influences on the phosphate that was released from diazinon photodegradation. When the natural photosensitizers were added, the amount of phosphate released increased significantly because of the diazinon indirect photodegradation by reactive species, such as the hydroxyl radical generated by NO3− and Fe3+. However, humic acid and HCO3− inhibited the phosphate released by a radical scavenging effect. When natural water was spiked with diazinon, the phosphate that was released in natural water was higher than that of the control or deionized water, and the phosphate that was released was inhibited when isopropanol was added to the reaction. In addition, the formation of hydroxyl radicals (˙OH) in the natural water systems was identified from the photoluminescence spectra using coumarin as the trapping molecule, and the steady-state concentration of ˙OH in natural water was 3.07 ± 0.57 × 10−16 M under UV irradiation. All of these results indicated that direct and indirect photolysis degradation of organic phosphorus significantly impacts the release of phosphate in surface waters.

 Please wait while we load your content...
Please wait while we load your content...