Mild synthesis of triarylsulfonium salts with arynes†
Abstract
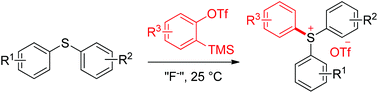
Reactions between arynes and alkyl sulfides have been extensively studied over the past few decades. These reactions commonly end with a dealkylation process and thus deliver thioethers as final products. In contrast, the transformation described furnishes valuable triarylsulfonium salts, in lieu of thioethers, from arynes and diarylsulfides. The reaction features mild conditions and a broad substrate scope. A suite of functional groups such as ketones, esters, nitriles, aryl ethers and aryl halides is tolerated, which can be issues faced by traditional synthetic methods. The practicality of the reaction and its extension to the synthesis of triphenyl selenonium salt are also exhibited herein.


 Please wait while we load your content...
Please wait while we load your content...