Refinement of labile hydrogen positions based on DFT calculations of 1H NMR chemical shifts: comparison with X-ray and neutron diffraction methods†
Abstract
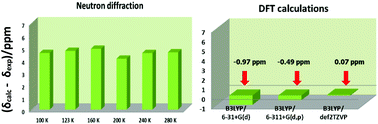
Numerous gas phase electron diffraction, ultra-fast electron diffraction, X-ray and neutron diffraction experiments on β-dicarbonyl compounds exhibiting enol–enol tautomeric equilibrium, with emphasis on acetylacetone and dibenzoylmethane, have so far been reported with conflicting results on the structural details of the O–H⋯O intramolecular hydrogen bond and resulted in alternative hypotheses on the intramolecular hydrogen bond potential function either a double minimum potential corresponding to two tautomeric forms in equilibrium or a single symmetrical one. We demonstrate herein, firstly, that the DFT calculated OH 1H NMR chemical shifts of acetylacetone and dibenzoylmethane exhibit a strong linear dependence on the computed O⋯O hydrogen bond length of ∼−50 ppm Å−1 and as a function of the O–H⋯O bond angle of ∼1 ppm per degree, upon the transfer of the hydrogen atom from the ground state toward the transition state. Secondly, the refinement of labile hydrogen atomic positions in intramolecular hydrogen bonds based on the root-mean-square deviation between experimentally determined and DFT calculated 1H NMR chemical shifts in solution can provide high resolution structures of O–H and O(H)⋯O bond lengths and O–H⋯O bond angles with an accuracy of ∼10−2 Å and ∼0.5°, respectively. Thirdly, the calculated 1H NMR chemical shifts in solution of the two ground state tautomers in equilibrium of acetylacetone and dibenzoylmethane are in excellent agreement with the experimental value, even for moderate basis sets for energy minimization. In contrast, the single symmetrical structure in a strongly delocalized system is a transition state with calculated 1H NMR chemical shifts which strongly deviate from the experimental value. Fourth, the DFT calculated ground state O–H bond lengths of acetylacetone and dibenzoylmethane are in quantitative agreement with the literature data which take into account the effect of quantum nuclear motion. The DFT structural results are critically discussed with respect to the state-of-the-art variable temperature X-ray and neutron diffraction methods.

 Please wait while we load your content...
Please wait while we load your content...