Theory of the thermodynamic influence of solution-phase additives in shape-controlled nanocrystal synthesis†
Abstract
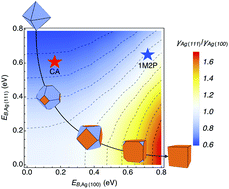
Though many experimental studies have documented that certain solution-phase additives can play a key role in the shape-selective synthesis of metal nanocrystals, the origins and mechanisms of this shape selectivity are still unclear. One possible role of such molecules is to thermodynamically induce the equilibrium shape of a nanocrystal by altering the interfacial free energies of the facets. Using a multi-scheme thermodynamic integration method that we recently developed [J. Chem. Phys., 2016, 145, 194108], we calculate the solid–liquid interfacial free energies γsl and investigate the propensity to achieve equilibrium shapes in such syntheses. We first apply this method to Ag(100) and Ag(111) facets in ethylene glycol solution containing polyvinylpyrrolidone (PVP), to mimic the environment in polyol synthesis of Ag nanocrystals. We find that although PVP has a preferred binding to Ag(100), its selectivity is not sufficient to induce a thermodynamic preference for {100}-faceted nanocubes, as has been observed experimentally. This indicates that PVP promotes Ag nanocube formation kinetically rather than thermodynamically. We further quantify the thermodynamic influence of adsorbed solution-phase additives for generic molecules, by building a γsl ratio/nanocrystal shape map as a function of zero-temperature binding energies. This map can be used to gauge the efficacy of candidate additive molecules for producing targeted thermodynamic nanocrystal shapes. The results indicate that only additives with a strong facet selectivity can impart significant thermodynamic-shape change. Therefore, many of the nanocrystals observed in experiments are likely kinetic products.


 Please wait while we load your content...
Please wait while we load your content...